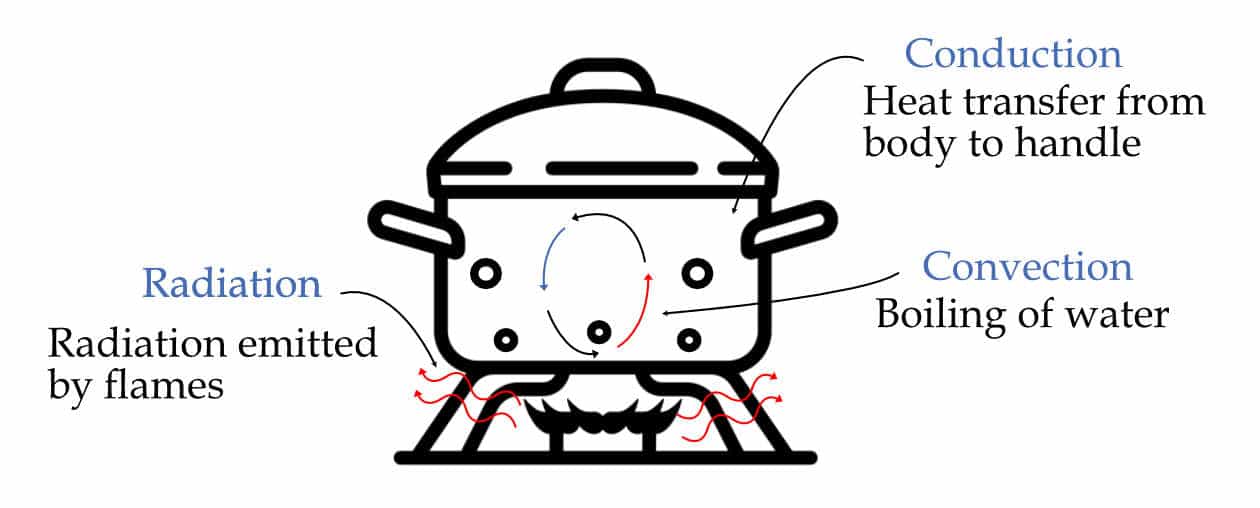
Heat transfer is the exchange of energy between two systems maintained at different temperatures. It is a boundary phenomenon.
Have you ever wondered why a hot cup of tea is spread on a saucer to cool it down, or a cold canned drink outside a refrigerator gets warmed up? The answer to these questions lies in heat transfer physics, which is the transfer of energy from a warm medium to a cold one.
Heat transfer is the exchange of thermal energy between two systems maintained at different temperatures. It is the science that deals with the rate of that energy transfer.
Heat v/s Thermal energy
Heat is a form of energy in transit that flows from a system at a higher temperature to a system at a lower temperature. Thermal energy, on the other hand, is the energy the system contains in molecular movements or vibrations. That is why in thermodynamics, you must have read that heat is a boundary phenomenon because it arises when there is an energy transfer from the system's boundary.
To summarize, the energy that transfers from one system to another is heat. And thermal energy is the energy stored in the system as its kinetic energy.
For example, when you are making tea on a stove, the transfer of energy from fire to the container and from the container to the tea mixture is termed heat. In contrast, the total amount of energy stored in the form of molecular vibrations in the tea is considered thermal energy.
Relation between Heat Transfer and Thermodynamics
Heat transfer and thermodynamics are two complementary branches of thermal science. Thermodynamics is the science focusing on the relationship between energy and work. And it includes dealing with the feasibility of any process for a particular system. On the other hand, heat transfer is the study of the rate of energy transfer from a system. In simple terms, thermodynamics studies the amount of heat transferred, whereas heat transfer studies the rate.
Heat always flows from a system at a higher temperature to a system at a lower temperature. But the mode through which the heat transfers can be different based on the nature of the medium or the presence or absence of a medium.
Broadly there are three modes:
Conduction: The transfer of energy from a more energetic particle of a system to adjacent less energetic ones of the same or different medium through physical interactions.
Convection: The transfer of energy due to the movement of the fluid (liquid or gas) particles between areas of different temperatures.
Radiation: The transfer of heat due to the movement of the fluid (liquid or gas) particles between areas of different temperatures.
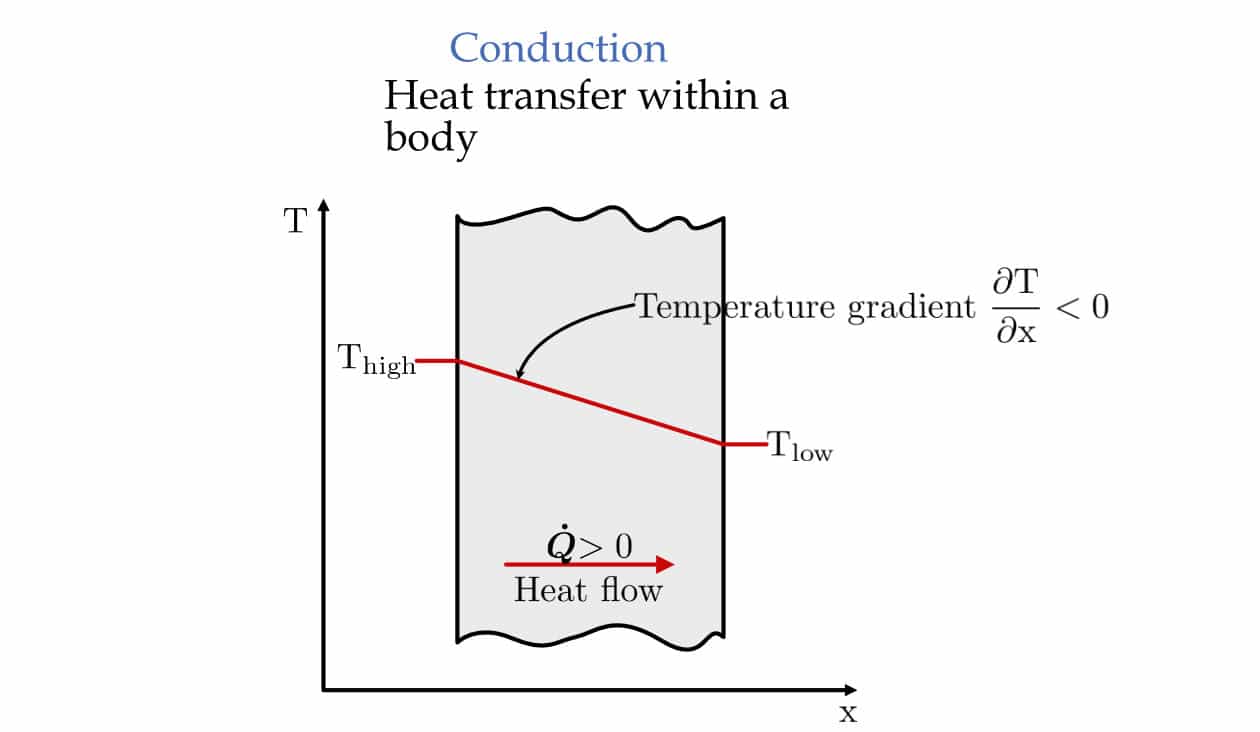
Conduction is the transfer of energy from a more energetic particle of a system to adjacent less energetic ones of the same or different medium through physical interactions. Simply put, when one energetic particle touches another with less energy, it will transfer some of its energy to another.
In engineering, conduction is often used to design heat exchangers, which transfer heat from one fluid to another. It is also important in the design of thermal insulation, which is used to keep heat from flowing into or out of a system.
This mode is present in all the major states of matter, i.e., solid, liquid, and gas. But how it is happening is different. In gases and liquids, conduction occurs through collision and diffusion of the molecules with random motion. In solids, it can be through lattice vibrations and energy transport by free electrons.
Fourier’s law of conduction
Joseph Fourier concluded that the heat transfer rate through conduction from a medium depends on the medium's geometry, the medium's material, and the temperature difference. He formulates the empirical law, which states that :
Fourier’s law: heat flux (q) resulting from thermal conduction is directly proportional to the magnitude of the temperature gradient.
or
where q is the heat flux, and Q is the heat transfer rate.
The constant of proportionality k is the material's thermal conductivity, and \frac{dT}{dx} is the temperature gradient. And A is the cross-sectional area.
The above equation helps in defining the temperature variation within a material with the help of the heat conduction equation.
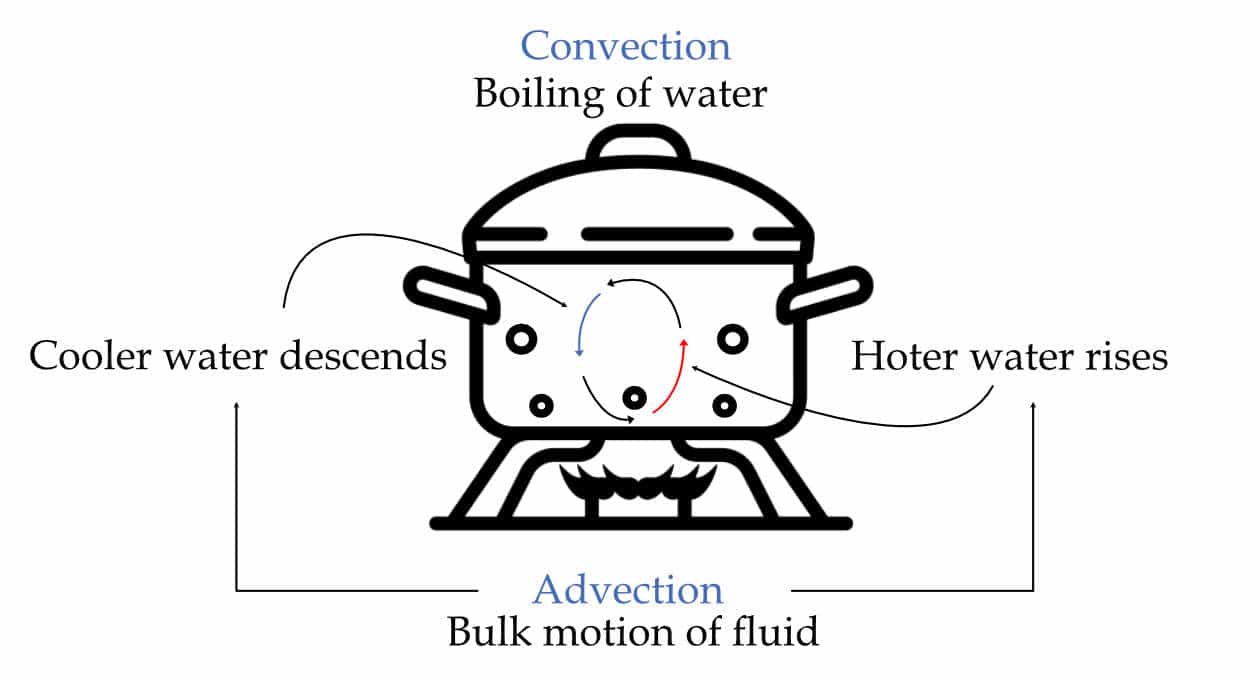
Convection is the transfer of heat due to the movement of the fluid (liquid or gas) particles between areas of different temperatures.
Conduction (Heat transfer) + Advection (Bulk motion) = Convection
It is a combination of two processes: conduction, in which heat transfer occurs between adjacent particles in the fluid. And advection, in which bulk motion of the liquid. Therefore, when there is no bulk motion, convection becomes pure conduction.
Thus, this mode of heat transfer is commonly used in heating, ventilation, and air conditioning (HVAC) systems, as well as in heat exchangers and boilers.
Newton’s law of cooling
Sir Isaac Newton formulated a law that defines the rate of cooling as a result of convection, known as Newton's law of Cooling. As per the law, the rate of heat transfer from the system is directly proportional to the temperature difference between the system and its surrounding.
or
where Q is the heat transfer rate, h is the heat transfer coefficient, and A is the area of the object.
Radiation is the phenomenon of energy transfer from one body to another body in the form of electromagnetic waves (or photons).
Any object above 0K emits thermal radiation due to a change in the electronic configuration of atoms or molecules. And since the mode of heat transfer is through electromagnetic waves, radiation doesn’t require the presence of an intervening medium to transmit energy.
As a result, this mode of heat transfer is important in the design of solar thermal technologies, which convert radiant energy from the sun into electricity. Furthermore, this is also crucial in the design of thermal protection systems for spacecraft, which must withstand the intense heat of re-entry into the Earth's atmosphere.

Stefan-Boltzmann law
The relation between the total radiation emitted and the body's temperature was given by Josef Stefan and theoretically derived by Ludwig Boltzmann. The law states that; thermal radiation energy emitted by a blackbody emitter is proportional to the fourth power, its absolute temperature.
Where σ is the Stefan-Boltzmann constant (5.67 × 10-8 Wm-2K-4), Ts is the body temperature, and A is the area of the body.
The radiation heat transfer between body and surroundings then becomes
From this post, you can get the answer to intriguing questions like why a hot cup of tea is spread on a saucer to cool it or how a thermos flask keeps the liquid at the same temperature for a long time.
Heat transfer is everywhere; it is the energy transfer between bodies maintained at different temperatures. It arises to bring the systems into thermodynamic equilibrium.
Some key learnings from the post:
Modes of heat transfer: There are three modes of heat transfer, i.e., conduction, convection, and radiation.
Conduction: The transfer of energy from a more energetic particle of a system to adjacent less energetic ones of the same or different medium through physical interactions.
Convection: The transfer of energy due to the movement of the fluid (liquid or gas) particles between areas of different temperatures.
Radiation: The transfer of heat due to the movement of the fluid (liquid or gas) particles between areas of different temperatures.
Your trusted source for breaking down core engineering concepts using first-principles thinking.