Thermodynamic systems are regions in space whose thermodynamic properties are of interest. There are three types of thermodynamic system.
How can you define yourself: an open thermodynamic system or a closed one? The answer can be vague on the philosophical front, but on the physics front, the answer is pretty clear you are an open system. Most engineering applications require knowledge of thermodynamic systems. For instance, in the case of a pressure cooker, you don't want it to be an open system during the cooking process, but you want it to be an open system when pressure is high.
You will learn in this post what is a thermodynamic system and what defines a system as open, closed, or isolated.
Thermodynamic systems are "regions in space whose thermodynamic properties are of interest". It is the part of the universe that is separated from the surrounding by real or hypothetical boundaries. The surrounding contains everything other than the system, including other thermodynamic systems, as shown in the figure below. In simple terms:
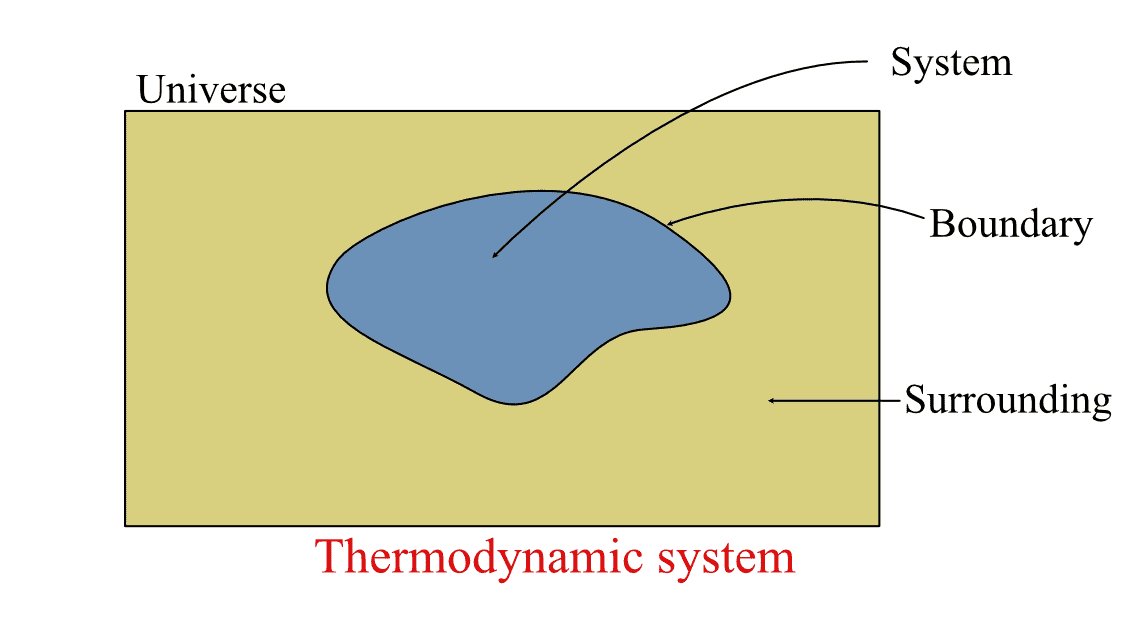
Thermodynamic system, and the boundary separating it from the surrounding,
The nature of its wall characterizes any thermodynamic system. These walls can be purely hypothetical when describing them as permeable to all matter, radiation and energies. Or it can be real, which is made of some matter.
The boundary of any thermodynamic system can be classified into these two types based on their ability to move:
Fixed boundary: When the system's boundary can not move and remains rigid.
Movable boundary: When the boundary of the system can move.
Other than the above classification, the boundaries can also be of these two types:
Real boundary: A tangible separating wall that separates the system from the surrounding.
Imaginary boundary: There is no real separation between the system and surroundings; there is just a hypothetical wall separating the system from the surroundings.
As described above, thermodynamic systems are the regions in space that are separated by boundaries. And the nature of these boundaries defined how mass and energy transfer across the thermodynamic systems and boundaries.
Every thermodynamic system in the universe can be classified into these three types:
Open system: If the thermodynamic system can exchange both matter and energy with its surrounding.
Closed system: If the thermodynamic system can exchange only energy with its surrounding.
Isolated system: If the thermodynamic system can neither exchange matter nor energy with its surrounding.
In the table and figure below, you can see which system allows energy and which doesn't:
| Type of system | Matter | Energy |
|---|---|---|
| Open system | Yes | Yes |
| Closed system | No | Yes |
| Isolated system | No | No |
Thermodynamic systems
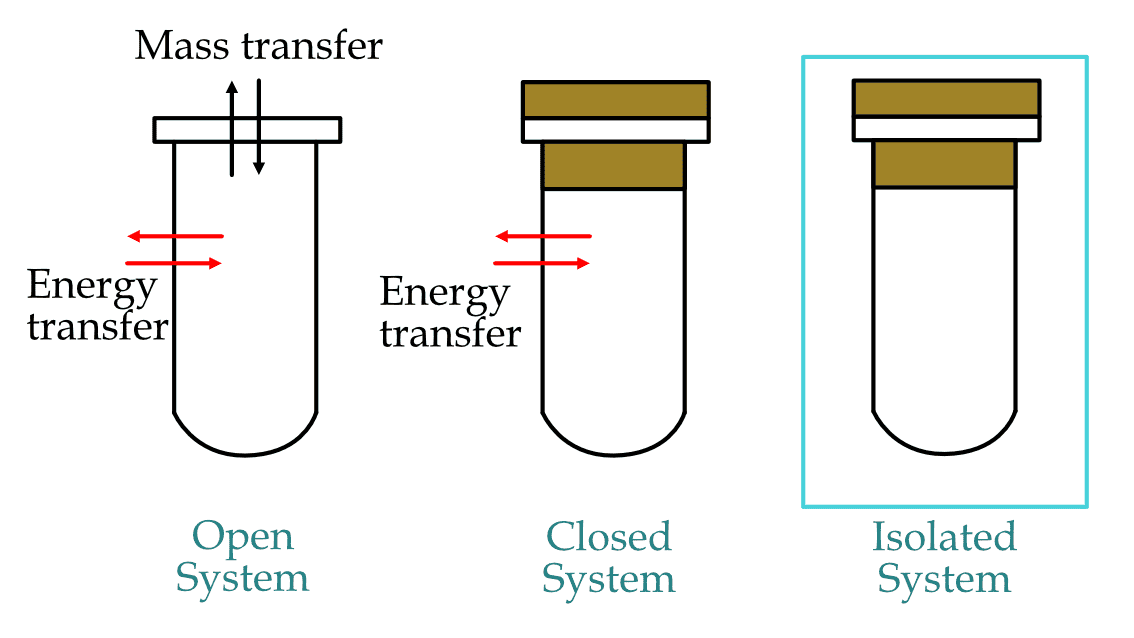
Types of thermodynamic systems
In an open system, matter and energy can transfer between the system and the surroundings. For such systems, the boundary that separates the system and surroundings allows matter and energy to pass through it.
For example, in tea in a cup, the vapors of heat can go outside the cup, and the heat is transferred through its boundaries. Another example is the human body; we are an open system because we take energy in the form of radiation and also exchange heat with our surrounding through our skin. And to survive, we eat, i.e., the matter enters and exits our body.
A closed system can only exchange energy with the surroundings and can not transfer matter through its boundaries. For such a system, the mass of the system remains constant. And the boundary is real, but it can be movable or rigid.
For example, when the cap is present in the water bottle, it behaves like a closed system.
A system can also change its type with time. For example, in the case of car or bike engines, when fuel enters into the piston-cylinder arrangement, then we can say that the piston-cylinder arrangement is an open system. But the same system becomes a closed system when the piston is moving for forwarding and backward stroke.
Isolated systems are those systems in which neither matter nor energy can transfer from the system. For these systems, the total energy remains constant.
For example, the universe is the most common example, where the energy remains constant.
Thermos-flask can also be considered an isolated system if it is completely insulated.
Thermodynamic systems are everywhere. And anything can be considered a thermodynamic system if the matter inside it is of our interest. Eventually, based on the nature of the boundaries, we classify the thermodynamic systems.
Some key learnings from the post:
Thermodynamic system: These are regions in space whose thermodynamic properties are of interest
Types of boundaries: Boundaries of any thermodynamic system can be real or imaginary and fixed and movable.
Types of thermodynamic systems: There are three thermodynamic systems, i.e., open, closed, and isolated.
Open system: The system can exchange matter and energy with its surroundings.
Closed system: The system can exchange only energy with its surrounding.
Isolated system: The system can neither exchange matter nor energy with its surrounding.
Your trusted source for breaking down core engineering concepts using first-principles thinking.