In nature, there are four fundamental forces. And every force in nature is nothing but one of these four fundamental forces. Find out how!
In nature, there are four fundamental forces: gravitational force, electromagnetic force, strong nuclear force, and weak nuclear force. But the question comes, “How can we classify any force into these four fundamental types ?”. It gets interesting when you try to answer that by going close to atomistic and molecular levels.
In this blog post, you will learn how the contact and non-contact forces are just different names for four fundamental forces.
For example, you are pushing your hands against the wall. In general, we say you are applying a contact (compressive) force on the wall. But, when you zoom in on a small portion where there is contact between the wall and the hand. Then, you can notice that what exactly is happening is that the atoms (nuclei) in your hands are pushing against the atoms (nuclei) in the wall since the nuclei are positively charged, therefore. As a result, there is electric repulsion between the nuclei, as shown in the figure below. So, it is a contact compressive force in the bigger picture, but in the small picture, it is just an electromagnetic force (electric and magnetic). And it is funny to say there is contact force because technically, atoms in your hands aren’t touching atoms in the wall.
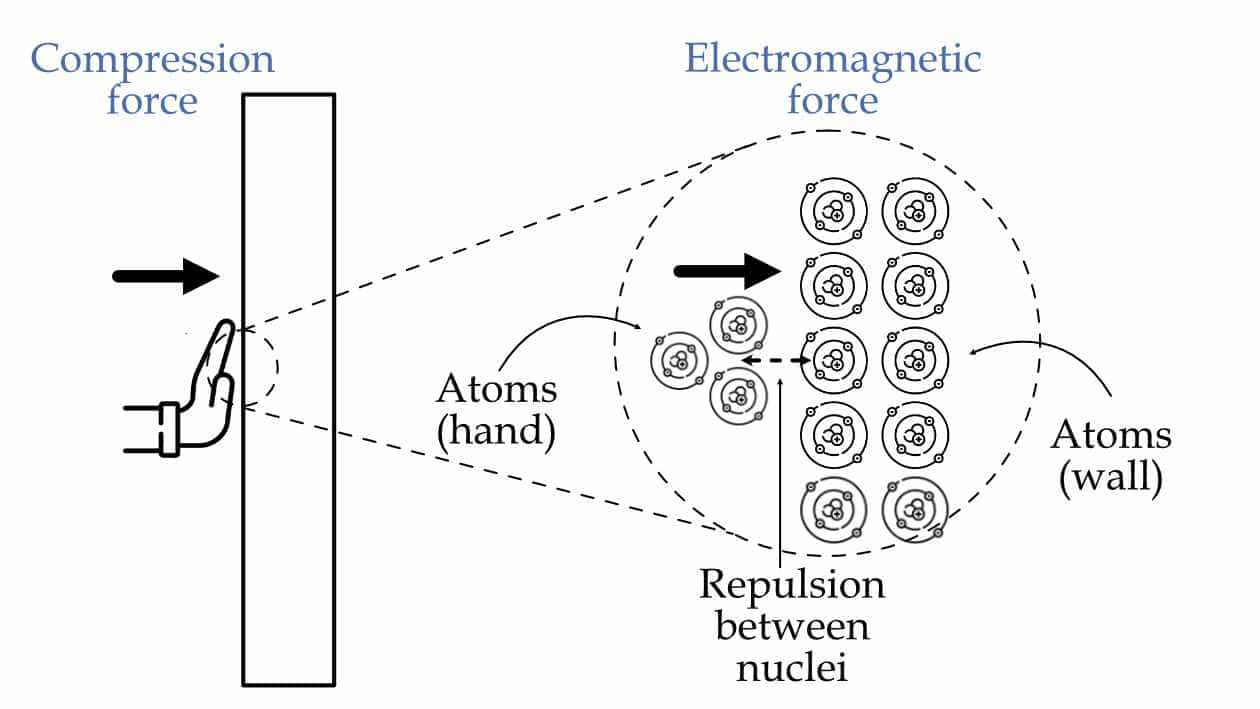
The compressive force acting on the wall is just an electromagnetic force acting between atoms of hand and wall.
Similarly, when you are pulling any wire with your hands. Then on the macroscopic level, you can say that you are applying tensile force on the wire. But when you zoom in to the microscope level, you can see that what exactly is happening is you are trying to pull atoms (or molecules) away from each other. As a result, there is a force of pull between the atoms to bring them down to their original position, as shown in the figure below. So, at the macroscopic level, it may look like a tensile force, but actually, it is just an electromagnetic force (pull).
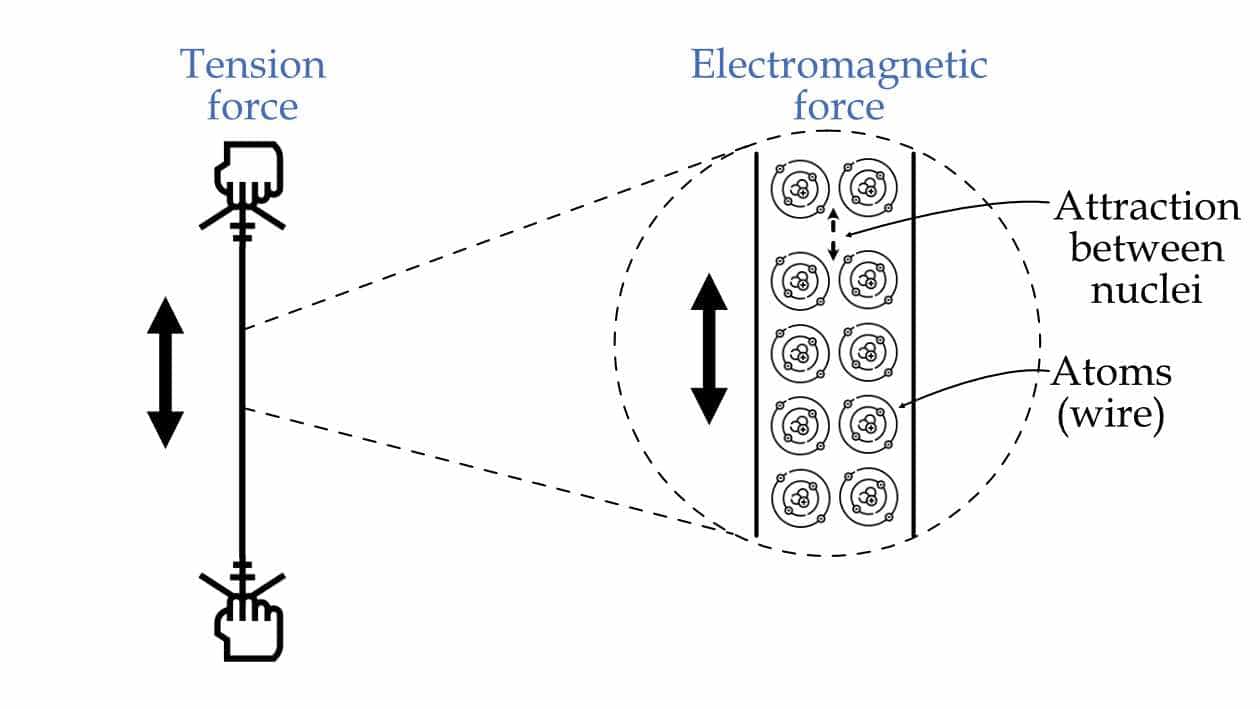
The tension force in the wire is the electromagnetic force between the wire's atoms.
You have seen examples that are closely related to mechanical forces. But, this concept of fundamental forces holds for all forces, including chemical interactions and bondings.
In hydrogen bonding, one molecule's partially negatively charged oxygen attracts the partially positively charged hydrogen of the same or different molecule. And the reason for that attraction is the pull between the oppositely charged particles. So, even hydrogen bonding is nothing but an electromagnetic interaction or force.
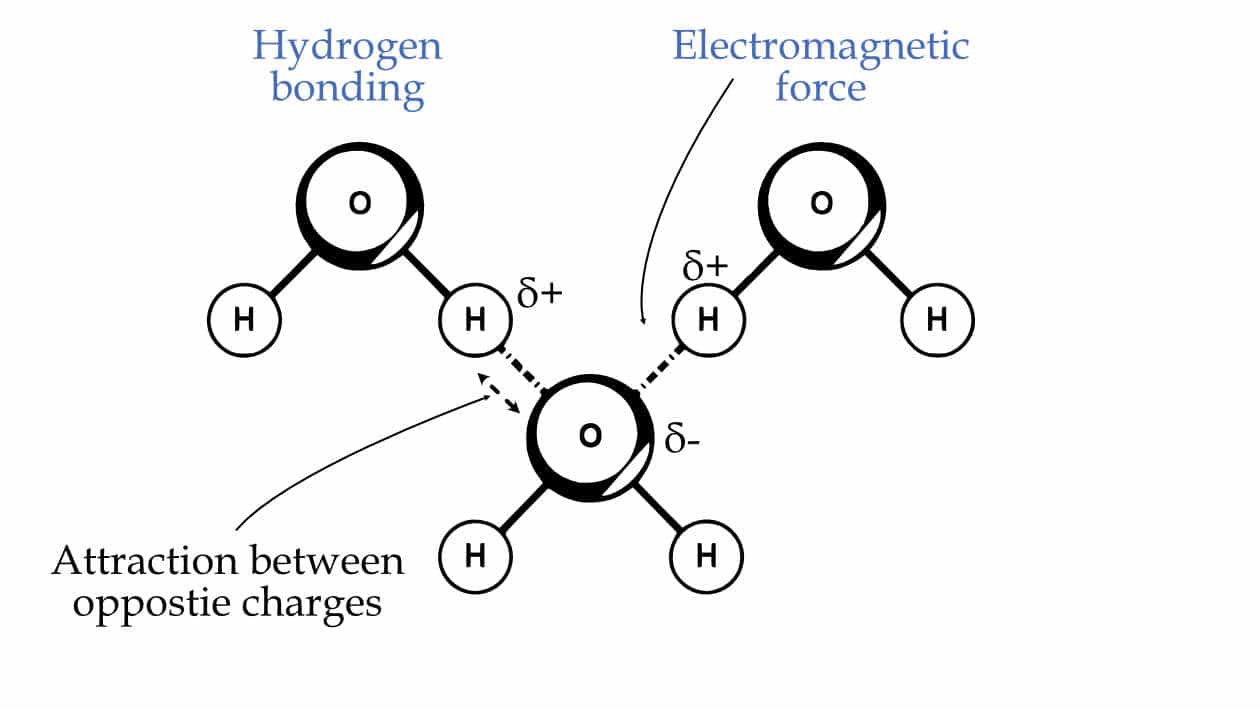
Hydrogen bonding is an electromagnetic interaction between partially negatively charged oxygen and positively charged hydrogen.
Nobel Laureate Richard Feynman has beautifully described this concept of the fundamental nature of the forces when answering the question "WHY ?"
Every force in nature is nothing but one of the four fundamental forces of nature. In a bigger picture it can be named either compression, tension or normal force. But, on a smaller picture it is just one of the four fundamental forces.
Some key learnings from this post:
Fundamental forces: In nature, there are four fundamental forces. And every other force is just derived from these four forces.
Every force is fundamental: Compressive, tensile, or chemical forces like hydrogen bonding are all fundamentally electromagnetic forces.
Your trusted source for breaking down core engineering concepts using first-principles thinking.