Learn basics about the isobaric process and its crucial role in maintaining performance in air conditioning, and internal combustion engines.
An isobaric process is a powerful concept in thermodynamics with far-reaching implications for many industries. From air conditioning and refrigeration to the operation of internal combustion engines, the isobaric process plays a crucial role in maintaining the efficiency and performance of these systems. Keeping the pressure constant, the isobaric process allows us to make precise calculations and predictions about how a system will behave.
In this blog post, you will learn the fundamentals of the isobaric process, along with real-world applications and mathematical formulations.
An isobaric process is a thermodynamic process in which the pressure of the system remains constant. In other words, during this process, the pressure of the system does not change despite changes in the system's properties.
This is in contrast with other types of thermodynamic processes, such as isothermal (constant temperature) and isochoric (constant volume). In an isobaric process, the pressure is constant and the volume may change. Mathematically this becomes:
Graphically, this process is represented with a straight line in the P-V diagram.
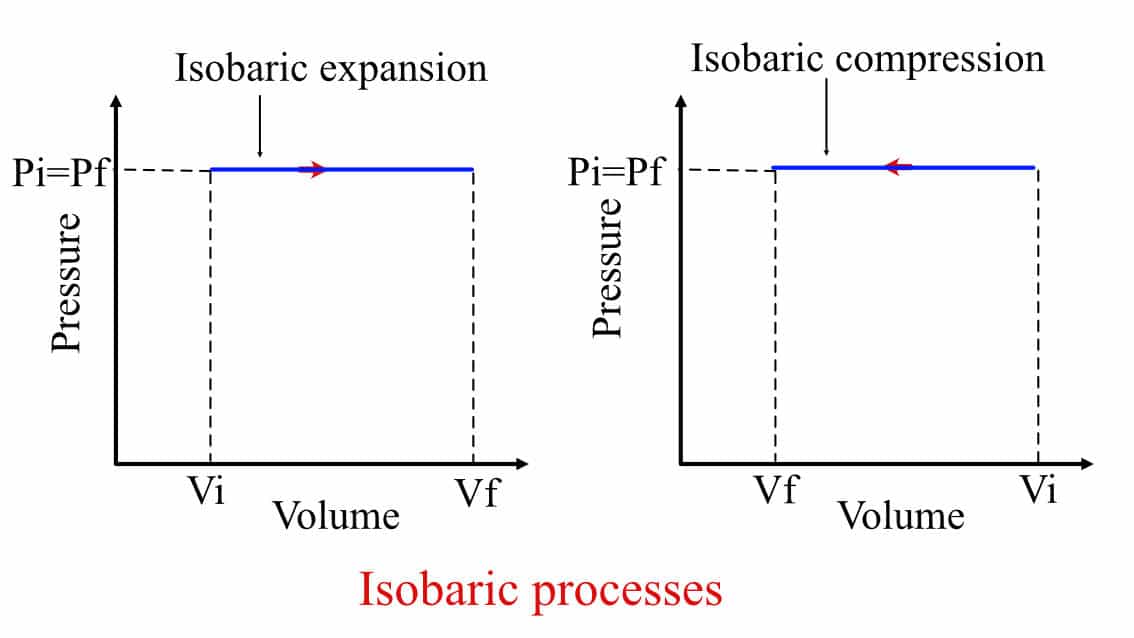
Isobaric expansion and compression process
In an isobaric process, the pressure of the system remains constant, so the work done by the system can be calculated using the equation:
Where P is the constant pressure, and ΔV is the change in the volume of the system during the process.
It's important to note that the work done in an isobaric process is the only case where the work done is directly proportional to the change in volume and not the pressure-volume product.
The work is represented in terms of P-dV work for a closed system undergoing this process. And for a system having ideal gas as the working substance, the work done becomes:
Where n is the number of moles, and R is the ideal gas constant. T is the constant temperature, V1 is the initial volume, and V2 is the final volume.
Graphically, it is the area under the isobars in the P-V diagram, as shown in the figure below. An isobar is any curve or contour having constant pressure throughout the process.
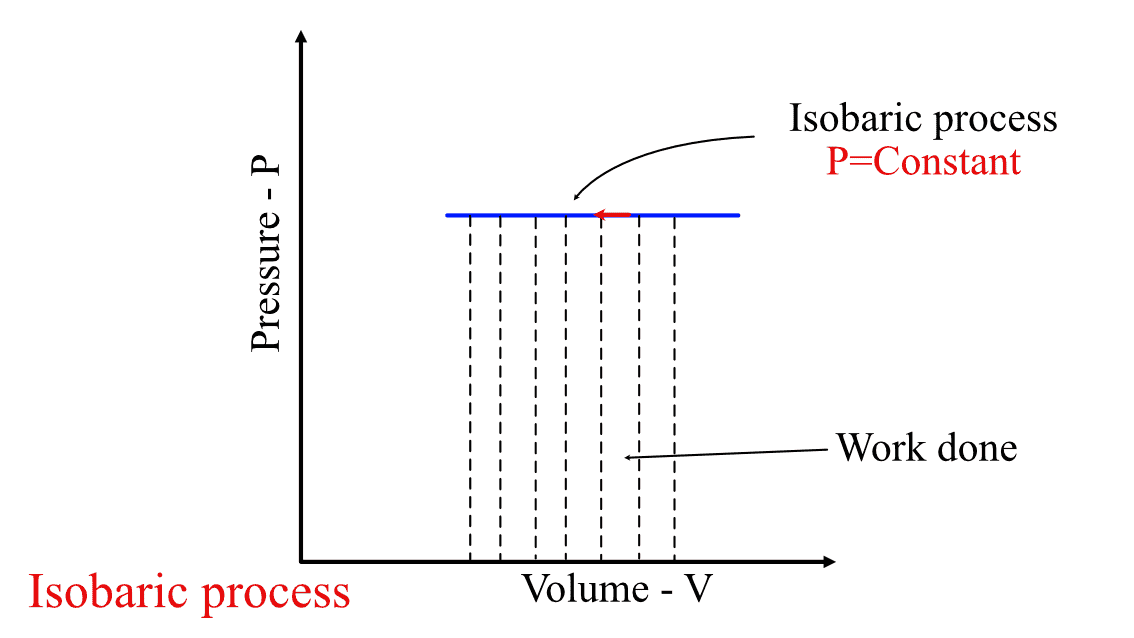
Work done in Isobaric process
The first law of thermodynamics is the fundamental law that defines the behavior of any system whether living or non-living. In the context of the isobaric process, there will be changes in the internal energy of the system, transfer of heat from the system, and work done by the system. Thus, no quantity in the first law of thermodynamics becomes zero.
For such a process, the first law of thermodynamics for a closed system becomes:
It's also worth noting that during such a process, the internal energy of the system can change (either increase or decrease) due to the addition or removal of heat, Q. The heat transfer during this process is where n is the number of moles, Cp is the heat capacity at constant pressure, and T1, T2 is the initial and final temperature.
In the kinematics of gases, the behavior of the ideal gases at constant pressure is well defined using Charle's law.
The law defined for constant pressure process for ideal gas states that "The volume of the gases is directly proportional to the absolute temperature of the fixed mass of the gas at constant temperature"
The law is named after Jacques Charles, who first formulated it in 1787. The mathematical expression of Charles's Law is:
where V is the volume of the gas, T is the absolute temperature of the gas, and k is a constant. This equation shows that when the pressure is constant, the volume of a gas will increase as its temperature increases, and decrease as its temperature decreases.
This process exists everywhere, from the ice melting in front of you to the heat transfer process taking place inside a jet engine. The applicability of these processes is diverse covering simple physics to complex engineering applications.
Some major engineering applications of the isobaric process are:
Air compressor: Air compressors compress air, raising its pressure and temperature. These have applications in powering pneumatic tools, providing breathing air for scuba diving, and operating air conditioning and refrigeration systems.
Jet engine and diesel engine: A jet engine is a type of internal combustion engine in which the combustion of fuel generates high-pressure and high-temperature gases that drives a turbine. And in a diesel engine, the heat addition takes place at constant pressure.
Heat pumps: A heat pump is a device that uses a small amount of energy to move heat from one location to another. It uses an isobaric process to transfer heat from the environment to a building or from one fluid to another.
Heat exchangers: The isobaric process can be used to transfer heat from one fluid to another without changing the pressure of either fluid
The isobaric process is a constant-pressure thermodynamic process that has got many practical applications such as in compressors, internal combustion engines, jet engines, heat exchangers, etc. These examples demonstrate the broad range of industries and systems in which this process plays a crucial role.
Some key learnings from the post:
Isobaric process: An isobaric process is a thermodynamic process in which the pressure of the system remains constant.
Work done: The work done during an isobaric process is : or
Heat transfer during the process: The heat transfer during an isobaric process is equal to
Practical applications: The isobaric process has many practical applications in various industries such as air conditioning and refrigeration, internal combustion engines, jet engines, heat exchangers, and heat pumps.
Charl's law: It states that "the volume of a gas is directly proportional to its absolute temperature at constant pressure"
Your trusted source for breaking down core engineering concepts using first-principles thinking.