The laws of thermodynamics are the fundamental rules applicable when a thermodynamic system undergoes some state change. The four laws are:
In this blog post, you will see the laws of thermodynamics and the practical significance of these laws.
The laws of thermodynamics are the fundamental rules applicable when a thermodynamic system undergoes some state change. These laws define the relationship between fundamental properties like temperature, energy, and entropy, which characterizes the state of a thermodynamic system at thermodynamic equilibrium.`
The laws of thermodynamics seem fairly easy to understand. But, their simplicity can often lead to underestimating the impact they have. Of the many aspects, they answer whether any process is feasible or not, if it is feasible, what is the optimal performance that you can get, and many more. The laws of thermodynamics apply to almost every aspect of scientific inequality in some way.
The four laws of thermodynamics are:
Zeroth law: If two systems are in thermodynamic equilibrium with a third system, then two original systems are also in thermal equilibrium.
First law: Energy can neither be created nor destroyed, but it can be changed from one form to another.
Second law: The entropy of an isolated system like the universe will always increase with time.
Third law: The entropy of a perfect crystal is zero at absolute zero temperature.
Zeroth law of Thermodynamics states that if two bodies are in thermal equilibrium with a separate third body, then the first two bodies are also in thermal equilibrium with each other.
This zeroth law of thermodynamics is a transitive property of thermal equilibrium. The transitive property of mathematics is that if A=B and B=C, then A=C. This same rule applies to thermodynamic systems in thermodynamic equilibrium.
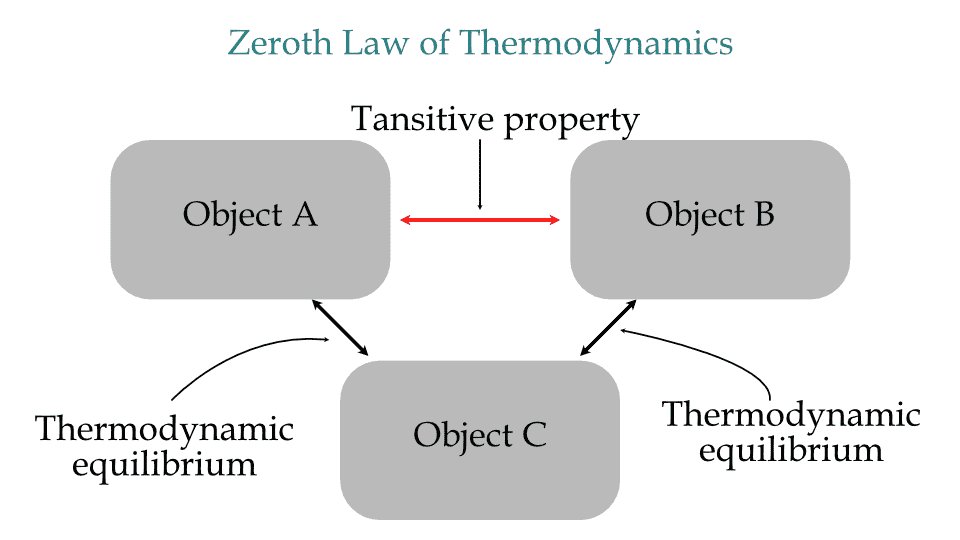
Zeroth law of Thermodynamics
For example, measuring the body's temperature using a mercury thermometer. The body is in thermal equilibrium with the thermometer metal tip, and the metal tip is in thermal equilibrium with the mercury inside. As a result, the body is in thermal equilibrium with mercury.
This law was understood without being explicitly stated throughout the history of thermodynamics. The law was formulated much after all the laws were formed. But since it was more fundamental than other laws, therefore it was named "Zeroth law"
The first law of thermodynamics, also known as the law of conservation of energy, states that energy can be converted from one form to another with the interaction of heat, work, and internal energy, but it can neither be created nor destroyed. Mathematically this law becomes:
where ΔU is the change in the system's total energy, Q is the heat exchanged between the system and surroundings, and W is the work done by the system.
Eventually, the consequence of this law is that the PMM1 (Perpetual Motion Machine 1) is not possible.
For example, automobile engines produce mechanical energy, but they convert the chemical energy of fuel into heat energy that is further converted into mechanical work.
The second law of thermodynamics states that the entropy of an isolated system will always increase over time. It fixates that the change in entropy of the universe can never be negative. Mathematically this law becomes:
where ΔS is the entropy (disorder within a system) change that defines the thermodynamic arrow of time. Hence, this law signifies whether any specific thermodynamic process, including the quasi-static process, will be practically possible or not.
Different scientists in different ways state the second law of thermodynamics. These are:
Clausius statement: It is impossible to transfer heat from lower to higher temperatures without external work.
Kelvin Plank statement: It is impossible to develop a device operating on a cycle that produces work with a single heat reservoir, i.e., efficiency can not be 100%.
Eventually, the consequence of this law is that the PMM2 (Perpetual Motion Machine 2) is impossible.
The third law of thermodynamics states that the entropy of a system approaches a constant value when its temperature approaches absolute zero, i.e., OK.
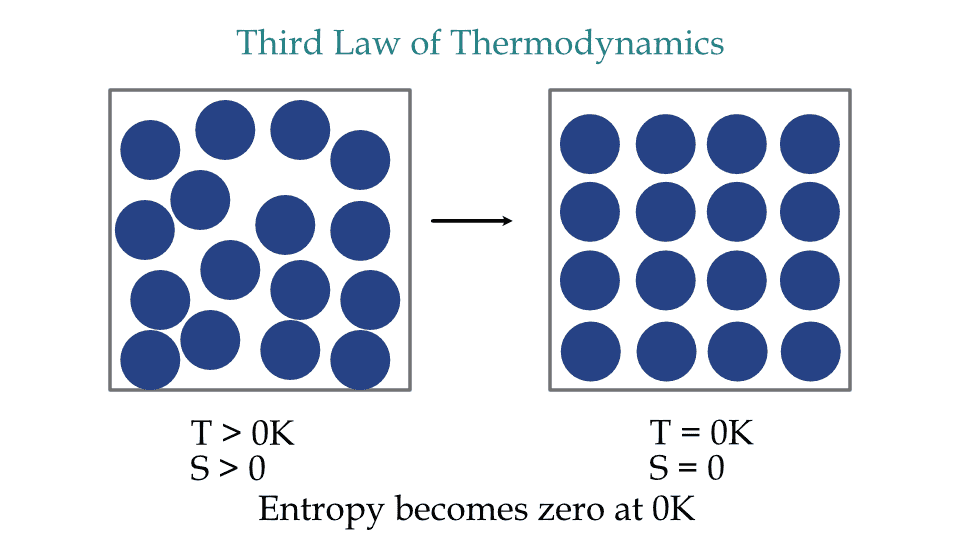
Third law of thermodynamics for a perfect crystal.
This law of thermodynamics relates the temperature and entropy of perfect crystals at the lowest possible temperature possible for any substances.
The third law of thermodynamics is stated by different scientists in different ways. These are:
Nernst theorem: It is impossible to reach the absolute entropy of a system to its absolute zero value in a finite number of steps.
Nernst-Simon statement: The entropy change of a system in a reversible isothermal process approaches zero as the temperature approaches zero.
Planck statement: In any system in equilibrium, the entropy tends to become constant, independent of the other thermodynamic variables, as the temperature approaches zero.
The major consequence of this law is that it is impossible to achieve absolute zero temperature, and at absolute zero temperature, the value of thermodynamics coefficients becomes zero.
Thermodynamic laws are much more widely applicable than you may even realize. The simplicity of these laws often leads to underestimating the impact they have. Whether measuring temperature with a thermometer or running a vehicle with fuel, thermodynamic laws are everywhere.
Some key learnings from this post:
Thermodynamic laws: The fundamental rules which are applicable when a thermodynamic system undergoes some sort of state change.
Zeroth law: If two systems are in thermodynamic equilibrium with a third system, then two original systems are also in thermal equilibrium.
First law: Energy can neither be created nor destroyed, but it can be changed from one form to another.
Second law: The entropy of an isolated system like the universe will always increase with time. This law tells whether any process is feasible or not.
Third law: The entropy of a perfect crystal is zero at absolute zero temperature.
Your trusted source for breaking down core engineering concepts using first-principles thinking.