A thermodynamic process is any process that involves a change in the thermodynamic state of the system. This is of five types.
Whether you are a mechanical engineer, chemical engineer, or any scientist. Your knowledge can always remain incomplete without thermodynamics or thermodynamic process. The reason is the science behind it, which is applicable everywhere, whether it's an automobile engine or some reaction in the lab.
Whenever there is a change in a thermodynamic system's state, some thermodynamic process is happening within the system. What is a thermodynamic process? What are the different types of processes and their work done? You will see that in this post.
Thermodynamic processes are any processes that involve changes in the thermodynamic state of the system.
The system evolves from one equilibrium microstate to another equilibrium microstate in a thermodynamic process. During that process, the system starts from an initial equilibrium state characterized by system parameters pressure (Pi), temperature (Ti), and volume (Vi), goes through various quasi-static states to a final equilibrium state characterized by system parameters pressure (Pf), temperature (Tf) and volume (Vf).
During these processes, heat or work may or may not transfer across the system boundaries.
Whenever any system undergoes a thermodynamic process, the properties (parameters) like temperature (T), pressure (P), volume (V), and internal energy (U) of the system change. For different processes, the variation of these properties follows some specific rules, and based on that, different processes exist. The governing equation or rule that defines the change of state of a system by the different processes can be different.
In thermodynamics, broadly, these major types of processes exist:
Isobaric process: The process in which pressure remains constant, i.e., ΔP =0.
Isochoric process: The process in which volume remains constant, i.e., ΔV =0.
Isothermal process: The process in which temperature remains constant, i.e., ΔT =0.
Adiabatic process: The process in which there is no heat transfer across the system boundaries throughout the process, i.e., Q =0. The system can be considered to be perfectly insulated. And as a result, in an adiabatic process, energy gets transferred only as work.
Polytropic process: Polytropic processes are any thermodynamic process in which the variation of pressure, volume, and temperature follow the PVn = C relationship. Here, n is the polytropic index.
The variation of pressure and volume for the different thermodynamic processes can be visualized with the PV diagram, as shown below.
Types of thermodynamic process
Learn about these processes in details here:
Isobaric process
Isothermal process
Isochoric process
Polytroic process
Isentropic process
For a closed system, the work done is represented as pressure-volume work that arises due to volume change in the system.
For any thermodynamic process acting on a closed system, the total work (W) done is
Where dW is the infinitesimal work done by the system. In the case of a reversible process, the work done by a closed system becomes a function of dV, where dV is the volume change of the system.
The work done by a closed system in the different thermodynamic processes are:
Isobaric Process
For this process, the pressure remains constant, and the system's volume changes. As a result, the work done (W) becomes:
So, in the case of expansion, the volume of the system increases; therefore, W is positive. And in the case of compression, the system's volume decreases; therefore, W is negative.
Isochoric Process
For the isochoric process, the volume remains constant throughout the process. Therefore, the work done becomes 0.
This process where there is no change in volume can be achieved only if the system boundaries are rigid.
Isothermal Process
For this process, the temperature remains constant. To find the work done for the isothermal process, consider the ideal gas equation PV = nRT. Using the ideal gas equation, P can be expressed in terms of temperature as P = \frac{nRT}{V}. Therefore, work done becomes:
So, in the case of expansion Vf is greater than Vi; therefore, W is positive. And in the case of compression, Vf is smaller than Vi; therefore, W is negative.
Adiabatic Process
For the adiabatic process, the heat transfer Q becomes 0, i.e., no heat is exchanged from the system. For this process, the relation between pressure and volume is PV_Ɣ_ = Constant. Thus, the work done becomes:
The application of the thermodynamic process is everywhere, whether device or engine. Whenever there is heat transfer or work transfer from a system, there can be some application of these processes.
For example, the automobiles like cars and bikes. On the very basic level, the petrol and diesel engine work on the Otto and Diesel cycle, respectively. And these cycles are nothing but a combination of the thermodynamic processes described above.
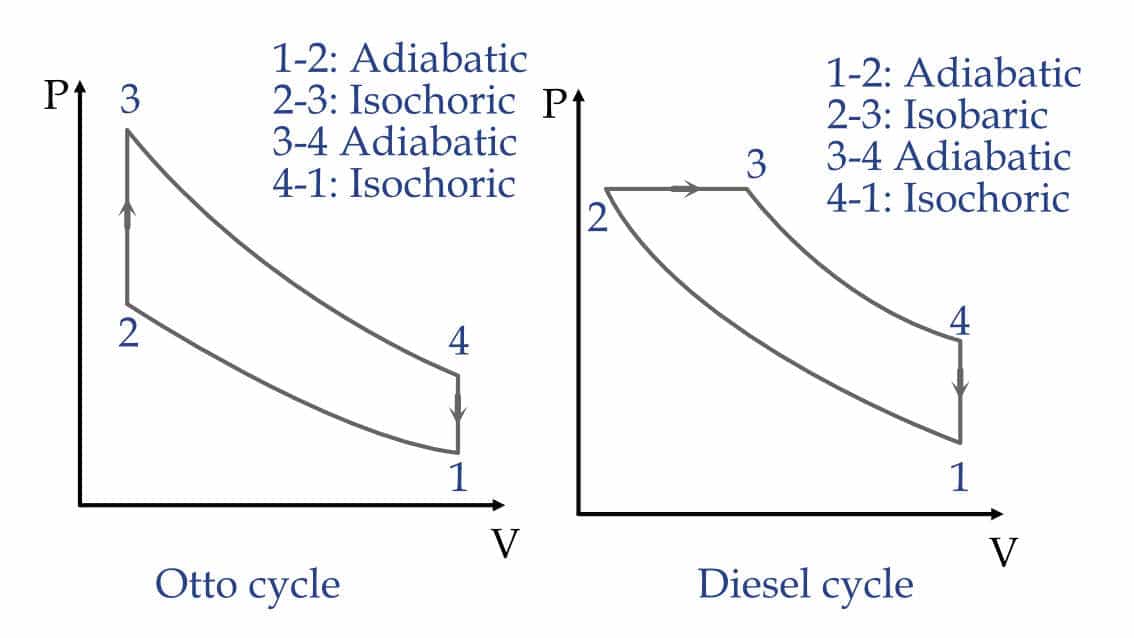
Graphical representation of Otto and Diesel cycle
Thus the knowledge of thermodynamic processes becomes the base for major thermal engineering applications.
The knowledge and understanding of the thermodynamic process are crucial for growing as an engineer or scientist. These processes are everywhere in nature, from the human body to the gigantic universe.
Key learnings from this post:
Thermodynamic process: Thermodynamic process are any process that involves a change in the thermodynamic state of the system.
Types of thermodynamic process: The thermodynamic process can be of five types, including isobaric, isochoric, isothermal, adiabatic, and polytropic processes.
Isobaric process: Process in which pressure remains constant throughout the process.
Isochoric process: Process in which volume remains constant throughout the process.
Isothermal process: Process in which temperature remains constant throughout the process.
Adiabatic process: Process in which there is no heat transfer across the system boundaries throughout the process.
Polytropic process: Polytropic processes are any thermodynamic process in which the variation of pressure, volume, and temperature follow PVn = 0 relationship.
Engineering application: Engineering applications of thermodynamic processes are everywhere, whether it's a car or bike engine or any chemical reaction.
Your trusted source for breaking down core engineering concepts using first-principles thinking.