Crack the Mysteries of Maxwell's Relations: a set of partial differential equations over a set of thermodynamic potential. Learn in details!
Thermodynamics is all about the behavior of systems and their related studies. The behavior of these systems is governed with the help of point functions that we call as fundamental thermodynamic quantities like pressure P, temperature T, volume V, and entropy S. And interestingly, these fundamental quantities aren’t independent but rather depend on each other. James Clark Maxwell pointed out this dependency on fundamental quantities and formulated the famous Maxwell’s relations.
This set of relations will help you in defining how the variation of one property affects the variation in other properties. Through this post, you will learn about these in detail.
Maxwell’s relations are a set of partial differential equations that are defined over a set of four fundamental thermodynamic potentials. These relations are derived using the symmetry of second derivatives when applied to the thermodynamic potentials.
As a result, we get the derived set of equations are:
here, , , , and are the thermodynamic potentials. And represents the internal energy, enthalpy, Helmholtz, and Gibbs free energy, respectively.
is temperature, is entropy, is pressure, and is volume.
By employing Maxwell's relations, scientists and engineers gain a deeper comprehension of the intricate connections between temperature, pressure, entropy, and other thermodynamic properties. These relations serve as powerful tools for investigating the behavior of complex systems and formulating equations that govern their dynamics. As we continue our exploration of thermodynamics, keep in mind the invaluable insights that Maxwell's relations offer and how they have revolutionized our understanding of the physical world.
The derivation process of Maxwell's relations requires the knowledge of differential equations and the basics of thermodynamics and thermodynamic potentials as the relations are born out of them.
Thermodynamic potential, also known as fundamental function, is a quantity that represents the state of a system. In total, we have four thermodynamic potentials those are internal energy , enthalpy , Helmholtz free energy , and Gibbs free energy .
Let's consider the first law of thermodynamics for a closed system in a differential form. Mathematically it is expressed as:
This equation is comparable to the differential equation of the form:
On comparing both the equations and applying the symmetry of derivatives, we can get Maxwell's first relation, i.e.
Similarly, the remaining relations can be derived using the same algorithm.
Now comes the question of how to remember these equations. Even though the whole idea of teaching is to ensure that learning is encouraged, not remembering, for exams, certain tricks (mnemonic) can help in swiftly recalling the relations.
One such trick is to consider 2nd and 3rd equations. Here you can see that it is T-V(Television) on the numerators and P-S (Play Station) in the denominators. And the subscripts are nothing but the denominators of the opposite terms.
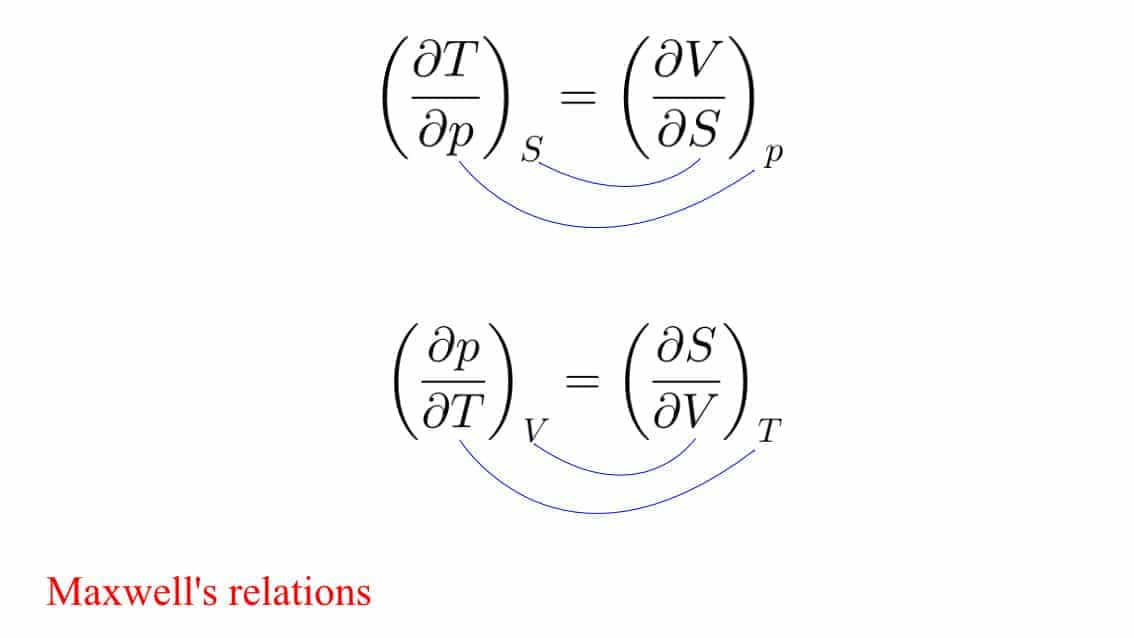
Maxwell's relations thermodynamics
Similarly, when you switch one term, then put a negative sign and then remodify the subscripts.

Maxwell's relations thermodynamics
Easy, right? You can also create your own ways.
In conclusion, Maxwell's relations offer an important foundation for comprehending how key thermodynamic quantities interact with one another. These relationships, derived from the symmetry of second derivatives, help formulate the equations that control the dynamics of complex systems and provide insightful information about how they behave. Scientists and engineers can better understand the physical universe by investigating the relationships between temperature, pressure, entropy, and thermodynamic potentials, including internal energy, enthalpy, Helmholtz free energy, and Gibbs free energy. Using mnemonic devices can also help you remember these equations for tests, enabling an easy recall of the relationships.
Key learnings from the post:
Definition: Maxwell's relations are a set of partial differential equations derived from the symmetry of second derivatives applied to thermodynamic potentials.
Derivation: The relations involve temperature, pressure, volume, entropy, and thermodynamic potentials like internal energy, enthalpy, Helmholtz free energy, and Gibbs free energy.
Application: Maxwell's relations offer valuable insights into complex systems and aid in formulating equations that govern their dynamics.
Remembering technique: Mnemonic techniques can help in remembering these equations for exams.
Your trusted source for breaking down core engineering concepts using first-principles thinking.