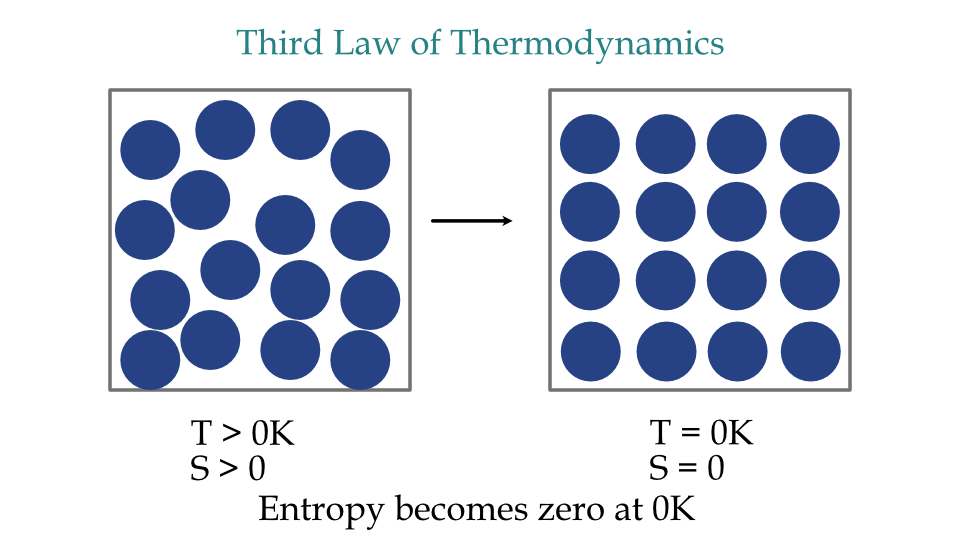
Third law of thermodynamics state that the entropy of a perfect crystal is zero when the temperature of the crystal is absolute zero.
The Third Law of Thermodynamics is like the "absolute zero" of thermodynamics and it's fascinating! It states that as the temperature approaches absolute zero, the entropy of a system reaches its lowest point. This principle has far-reaching implications in thermodynamics and is used in a wide range of practical applications in engineering. Imagine designing super-efficient refrigeration systems or understanding how materials behave at ultra-low temperatures, thanks to the Third Law.
This blog post will answer what the third law of thermodynamics is and where it is applicable.
The entropy of a system approaches a constant value when its temperature approaches absolute zero, i.e., 0 K.
Or in other words.
The entropy of a perfect crystal is zero when the temperature of the crystal is absolute zero, i.e., 0 K.
The third law of thermodynamics, also known as the Nernst law, can be defined as, on reaching the absolute zero temperature (0 K), any physical process stops; when any system reaches absolute zero temperature, the entropy reaches a minimum constant value.
The third law of thermodynamics deals with the behaviors of systems, particularly perfect crystals, as the temperature reaches absolute zero. This law of thermodynamics relates the temperature and entropy of perfect crystals at the lowest possible temperature possible for any substance. However, this definition only holds true for perfect crystals. In the case of real crystals, there will be inherent disorder in the crystals which will not be removed by cooling to absolute zero. As a result, the information needed to describe these defects will add to the entropy of the crystal. And it will not reach the minimum value.
As per statistical mechanics, the entropy of any system is a function of initial entropy, which is
where S and S0 are the entropy and initial entropy of the system and Kb is the Boltzmann constant. Ω is the total number of microstates in the system (the number of ways the system can become ordered).
For a perfect crystal, the number of microstates is 1. And on considering the initial entropy as zero, the entropy of the perfect crystal at absolute zero becomes
Thus, the entropy of a perfect crystal becomes zero at absolute zero temperature.
Third law of thermodynamics for a perfect crystal.
Different scientists postulated the third law of thermodynamics differently, describing the system's behavior at absolute zero temperature.
The important theorems and statements of the third law of thermodynamics:
Nernst theorem: It is impossible to reach the absolute entropy of a system to its absolute zero value in a finite number of steps.
Nernst-Simon statement: The entropy change of a system in a reversible isothermal process approaches zero as the temperature approaches zero.
Planck statement: In any system in equilibrium, the entropy tends to become constant, independent of the other thermodynamic variables, as the temperature approaches zero.
The third law of thermodynamics helps calculate the value of a substance's absolute entropy at any temperature (T). This relation between the entropy and temperature is based on the heat capacity of the substance.
For a solid, having S0 entropy at O K and S entropy at temperature T K. The value of entropy S is
Considering a perfect crystal having S0 = 0, the entropy becomes
The value of the integral can either be calculated graphically by plotting \frac{Cp}{T} or analytically by integrating the function. Thus, the value of entropy at any temperature T becomes
where Cp is the heat capacity of the substance at constant pressure.
The third law dealing with the behavior at absolute zero temperature describes some of the crucial points of the thermodynamic process.
The key consequences as implied by the third law of thermodynamics:
Absolute zero temperature can not be reached: The absolute zero temperature can not be achieved in any number of finite steps. It can only be approached.
Behavior of thermodynamic coefficients: As the temperature approaches zero, the value of heat capacity, coefficient of thermal expansion, and other thermodynamic coefficients reach zero.
The third law of thermodynamics is the law that describes the relationship between entropy and temperature for closed systems and perfect crystals at absolute zero temperature.
Some key learnings from the post:
Third law for perfect crystal: The entropy of a perfect crystal is zero when the temperature of the crystal is absolute zero.
Third law for the closed system: The entropy of a system approaches a constant value when its temperature approaches absolute zero.
Consequences of Third law: The absolute zero temperature can not be reached in finite steps. And the value of thermodynamic coefficients becomes zero as the temperature reaches absolute zero.
Your trusted source for breaking down core engineering concepts using first-principles thinking.